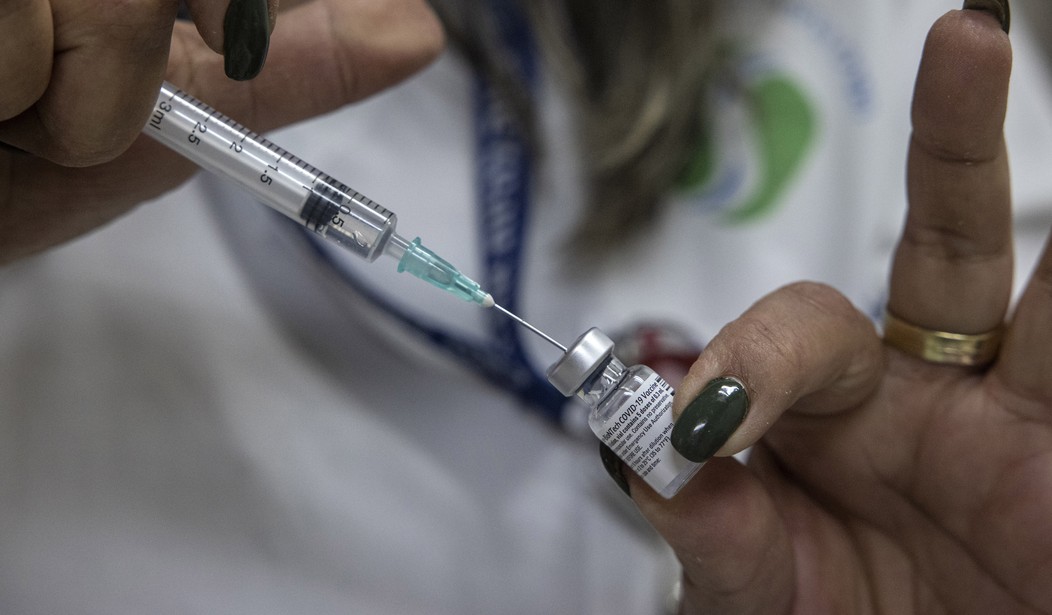
Get ready for the first wave of booster distribution. The Food and Drug Administration (FDA) has just issued an emergency authorization for the Pfizer vaccine.
From the Epoch Times:
The Food and Drug Administration on Wednesday authorized for emergency use a booster dose of the Pfizer-BioNTech vaccine for those aged 65 and older, as well as those aged 18–64 who are at high risk of severe COVID-19.
The third dose of the mRNA-based shot is authorized to be administered at least six months after the first two doses of the vaccine.
This decision was made after the FDA advisers decided that booster shots should only be issued for those over age 65 or for those who are in high risk categories. President Joe Biden had previously announced that booster shots would be administered to all Americans 16 years and older, but he received push back on this. Certain FDA scientists even resigned over what it perceived as added pressure from the White House to approve booster shots for everyone.
A third population eligible for the dose under the amended FDA emergency use authorization (EUA) are those aged 18-64 “whose frequent institutional or occupational exposure to SARS-CoV-2 puts them at high risk of serious complications of COVID-19 including severe COVID-19.”
This third category includes “health care workers, teachers, and day care staff, grocery workers, and those in homeless shelters or prisons,” among others, Acting FDA Commissioner Janet Woodcock said in a statement.
The Center for Disease Control’s Advisory Committee on Immunization Practices (ACIP) is expected to meet on Thursday to vote on proper administration and use of the booster. Once this vote is affirmed, the CDC will issue guidance on how best to distribute the Pfizer booster shot to the appropriate populations.
President Biden announced Monday that he plans to receive his booster shot on camera.











Join the conversation as a VIP Member