One of the more disturbing things to come from the COVID-19 crisis is the politicization of treatments and behaviors, framing things that could have positive health benefits in helping fight the virus as dangerous and unworthy of risk because Donald Trump has recommended them.
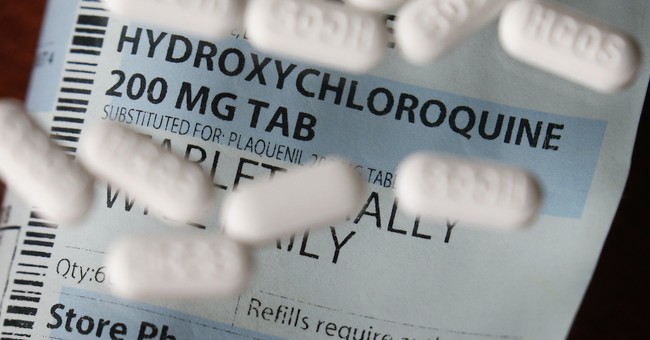
The most prominent has been the political battle over the use of hydroxychloroquine.
Trump initially touted the anti-malarial drug as a potential game changer, and the effort to prove him wrong has been afoot ever since; even as doctors across the nation have been touting its effectiveness (rather dramatically) and other nations across the world have used it as a prophylactic for physicians and those who’ve had contact with the infected almost from the start of the crisis.
Now, even as new headlines blare the drug simply doesn’t work and may even be fatal, the National Institutes of Health (NIH) will begin a clinical trial looking at if the drug, administered to those in the early stages of the COVID infection, can help slow its progression and keep patients from becoming critically ill and requiring hospitalization.
The Phase 2b trial will enroll approximately 2,000 adults at participating ACTG sites(link is external) across the United States. Study participants must have confirmed infection with SARS-CoV-2, the virus that causes COVID-19, and be experiencing fever, cough and/or shortness of breath. The investigators anticipate that many of those enrolled will be 60 years of age or older or have a comorbidity associated with developing serious complications from COVID-19, such as cardiovascular disease or diabetes. Participants will be randomly assigned to receive short-term treatment with either hydroxychloroquine and azithromycin or matching placebos. People living with HIV and pregnant and breastfeeding women also are eligible to participate in the study. The first participant enrolled today in San Diego, California.
“We urgently need a safe and effective treatment for COVID-19. Repurposing existing drugs is an attractive option because these medications have undergone extensive testing, allowing them to move quickly into clinical trials and accelerating their potential approval for COVID-19 treatment,” said NIAID Director Anthony S. Fauci, M.D. “Although there is anecdotal evidence that hydroxychloroquine and azithromycin may benefit people with COVID-19, we need solid data from a large randomized, controlled clinical trial to determine whether this experimental treatment is safe and can improve clinical outcomes.”
There has recently been a high-profile firing within the Trump administration related to the battle over the drug. Rick Bright, former director of the Biomedical Advanced Research and Development Authority, claimed whistleblower status and said “he was removed from his position in part because of his reluctance to promote the use of chloroquine and hydroxychloroquine, because they had not been tested and deemed safe for treating covid-19.”
Given that most of the studies claiming that the drug doesn’t work have been observational — meaning the drug was administered to patients (in some cases already very ill patients) and these patients were simply observed to see how they responded — a true double-blind, randomized clinical trial conducted by the NIH couldn’t have come at a better time.
And, honestly, should have come much sooner.










Join the conversation as a VIP Member